
Juvederm Ultra Smile (2x0,55ml)
Enhancing lip volume | Improving lip fullness and contour | Lifting the corners of the mouth | Emphasizing the Cupid's bow
Medical device. Contains lidocaine.



Reduction of sun and hormonal discolorations | Even skin tone | Melasma and chloasma
Non-crosslinked hyaluronic acid + biorevitalizing cocktail (whitening agents, flavonoids, vitamins, amino acids, trace elements, polyphenols) | medical device
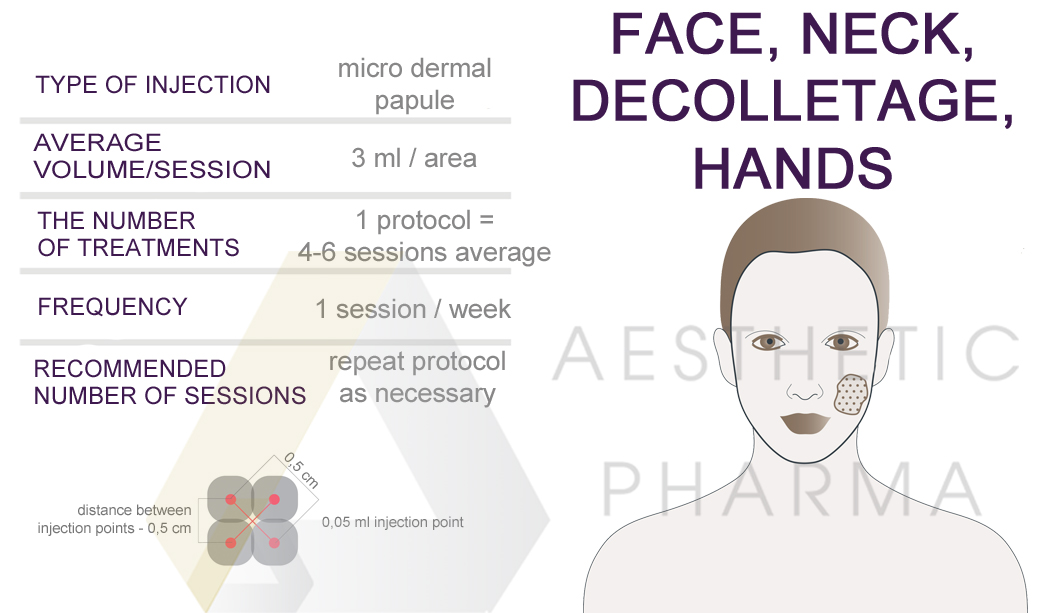
Application areas: Face | Neck | Décolleté | Hands

Privacy Policy

Shipping & delivery

Payment methods

RRS® Whitening is designed for skin regeneration and restoring its uniform color tone, such as discoloration from sun exposure or hormonal pigmentation. Hyaluronic acid in RRS® Whitening aims to hydrate the skin, while the biorevitalizing cocktail (including whitening agents) combats discolorations and restores a uniform, healthy appearance to the skin.
RRS® Whitening is a medical device Class III - CE 1014, intended for injection.
RRS® Whitening can be used on areas such as:
For whom: no age limit.
Injection technique: micro-deposit technique.
Recommended needle size: 32G.
Best results are achieved after 4-6 treatments performed once a week. This session can be repeated as needed.
Combination with other treatments:
0.1 mg/ml non-crosslinked hyaluronic acid + 65 mg/ml biorevitalizing cocktail
Price is for 1 vial of 3 ml preparation.
The whole package contains 6 vials of 3 ml each.
The procedure should be performed by a specialist familiar with the administration method.
The procedure should be carried out manually using the multi-point injection method or with a mesotherapy gun. RRS Whitening is indicated only for injections into the superficial or middle dermis using a 32G needle.
Before the procedure, patients should be informed about possible side effects and risks. The area to be treated should be thoroughly disinfected. The product should be injected slowly into the skin.
After the procedure, slight swelling, redness, or minor bruising may occur, which will disappear within a few hours to several days.
Store at temperatures from 2 to 30°C. Protect from sunlight and freezing. Do not freeze. Keep out of reach of children.
Skin Tech Pharma Group SL
Anti-aging | Regeneration | Improvement of tension - soft lifting effect
Non-crosslinked hyaluronic acid + revitalizing cocktail | Medical device
Application areas: Face | Neck | Décolleté | Belly button | Abdomen | Inner part of hands and thighs
Spłycenie drobnych zmarszczek | Miękki lifting skóry | Nawilżenie | Redukcja przebarwień | Biorewitalizacja
usieciowany kwas hialuronowy + bufor aminokwasowy | wyrób medyczny
Biostimulation of hair growth | Improvement of hair condition and structure | Scalp hydration | Hair loss
Non-crosslinked hyaluronic acid + biostimulating cocktail (FGF growth factor, amino acids, trace elements, vitamins) | medical device
Application areas: Scalp | Eyebrows | Pubic mound
Organic silicon - participates in the formation of collagen fibers in the skin | Improves skin tension | Moisturizes tired skin
Organic silicon + biorevitalizing cocktail (polyphenols, antioxidants) | medical device
Areas of application: Face | Neck | Décolleté | Hands | Navel | Abdomen | Belly | Inner part of hands and thighs | Knees
Reduction of stretch marks | Scar lightening | Firming body skin
Non-crosslinked hyaluronic acid + revitalizing cocktail (FGF growth factors, amino acids, trace elements, vitamins, coenzymes, antioxidants) | Medical device
Area of application: Buttocks | Thighs | Abdomen | Arms
Reducing Fine Lines | Hydration | Stimulation and Regeneration of the Skin
Non-crosslinked hyaluronic acid + revitalizing cocktail (vitamins, coenzymes, antioxidants, amino acids, microelements, polyphenols) | medical product
Application Areas: Face | Neck | Décolleté | Abdomen | Back of Hands | Inner Part of Hands and Thighs
Cellulite reduction | Local fat corrector | Firming and improving skin quality of the body
Non-crosslinked hyaluronic acid + revitalizing cocktail (caffeine and fucoxanthin, antioxidants, lipolytics, trace elements, vitamins, coenzymes, antioxidants) | medical device
Application areas: Chin | Buttocks | Thighs | Abdomen | Back | Arms
Moisturizing and skin regeneration around the eyes | Brightening the eye area | Improving microcirculation
Non-crosslinked hyaluronic acid + biorevitalizing cocktail (antioxidants, flavonoids, saponins, polyphenols, peptides, organic silicon) | medical device
Application area: Eye Area
Anti-aging | Superficial wrinkle correction | Soft lifting | Skin firmness improvement
non-crosslinked hyaluronic acid + biorevitalizing cocktail (amino acids, polyphenols, antioxidants) | medical device
Application areas: Face | Neck | Décolleté | Hands | Inner arms and thighs
Intensive skin moisturizing | Reduction of fine lines | Ideal for smoker's skin | Regeneration of mature skin
Uncrosslinked hyaluronic acid + oligopeptides | Medical device
Area of application: Face | Neck | Décolleté